Keeping up with healthcare and wellness news from the world
Provided by AGPBacteria may hold a cancer treatment clue
GA, UNITED STATES, May 15, 2026 /EINPresswire.com/ -- A research team has proposed a provocative shift in how some of the hardest-to-treat cancers might be understood and managed. The review argues that tumors with reproducibly poor prognosis outcomes may share an overlooked feature: bacterial infection within the tumor microenvironment. These bacteria may weaken treatment by altering local immunity, damaging tissue structure, and even inactivating chemotherapeutic drugs. Rather than waiting decades for more advanced nanodrugs to reach broad clinical use, the authors suggest that selected poor prognosis outcome (PPO) tumors may need to be treated from the outset as potentially bacteria-infected, using familiar carrier-free antibiotic and chemotherapeutic drug combinations to restore treatment sensitivity.
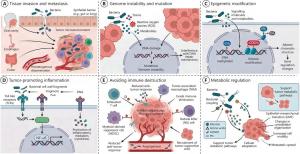
Cancer treatment has made major progress through targeted therapies, immunotherapies, improved diagnostics, and supportive care, turning several cancers into chronic conditions. Yet this progress has not reached all patients equally. Pancreatic ductal adenocarcinoma, colorectal carcinoma, and some biliary cancers often remain difficult to control because of rapid genetic evolution, immune suppression, poor drug penetration, hypoxia, fibrosis, and chemotherapy resistance. At the same time, intratumoral bacteria have been increasingly detected in multiple human cancers, but reliable clinical tools to diagnose tumor infection in living patients remain unavailable. Based on these challenges, there is a need to carry out in-depth research into bacterial infection as a clinically relevant feature of poor-prognosis tumors.
Researchers from Nankai University, the University of Utah, and Tianjin Medical University Cancer Institute & Hospital published this review in April 2026 in Cancer Biology & Medicine. The article examines whether bacteria within poor prognosis outcome tumors may help explain persistent treatment resistance, why nanodrug delivery has shown limited clinical benefit despite strong preclinical promise, and whether early carrier-free antibiotic–chemotherapeutic combinations could offer a faster, clinically practical route to re-sensitize difficult-to-treat tumors.
The review builds its argument across oncology, microbiology, and nanomedicine. First, the authors summarize evidence that bacteria are found in several poor prognosis outcome (PPO) tumors, including pancreatic ductal adenocarcinoma, colorectal carcinoma, and biliary tract cancers. Some species, such as Fusobacteriumnucleatum, Escherichia coli, Enterococcus faecalis, and Pseudomonas species, have been linked to immune evasion, inflammation, chemoresistance, or drug metabolism. Second, the authors examine why nanoparticulate chemotherapeutics have not yet delivered the expected clinical leap. Although nanoparticle delivery often appears stronger in rodent models, human tumors are more heterogeneous, fibrotic, and structurally complex. The enhanced permeability and retention (EPR) effect, often used to explain nanodrug accumulation in tumors, may therefore be less reliable in patients than in animal models. Third, the review evaluates three hypotheses: all diagnosed PPO tumors should be considered bacteria-infected; these tumors may show infection-related enhanced permeability to chemotherapeutics; and initial treatment should include carrier-free antibiotic and chemotherapeutic combinations. Evidence from animal models suggests that adding antibiotics such as ciprofloxacin may help re-sensitize infected tumors to chemotherapeutics such as gemcitabine.
The authors said the central message is not that nanomedicine lacks promise, but that time matters differently for patients with PPO tumors. They said advanced nanocarriers may ultimately improve tumor targeting, yet their clinical optimization could take decades. For patients facing cancers with short treatment windows, a more immediate strategy may be needed. They argue that clinically familiar antibiotic–chemotherapeutic combinations deserve serious retrospective evaluation, especially where bacterial infection is plausible but difficult to diagnose. This approach, they said, asks whether existing drugs can be used more intelligently before new platforms are ready.
The proposed paradigm has important clinical and translational implications. Treating PPO tumors as presumptively bacteria-infected would require careful antibiotic stewardship, because unnecessary antibiotic use can disrupt protective microbiomes and contribute to resistance. However, many patients receiving chemotherapy already require antibiotics because of infection risks related to immune suppression, catheters, or biliary stents. The authors therefore suggest that carefully selected carrier-free antibiotic and chemotherapeutic combinations could offer a practical near-term path to test whether bacterial involvement contributes to treatment failure. If supported by retrospective clinical data and future studies, this strategy may help reshape treatment planning for cancers that remain resistant to current standard approaches.
References
DOI
10.20892/j.issn.2095-3941.2025.0748
Original Source URL
https://doi.org/10.20892/j.issn.2095-3941.2025.0748
Funding information
D.Y.W. acknowledges financial support from the National Key R&D Program of China (Grant No. 2022YFA1205700), the National Natural Science Foundation of China (Grant Nos. 52503184 and 52293383), the China Postdoctoral Science Foundation (Grant Nos. 2023T004TJ and 2024M751517), and the Tianjin Key Projects of Public Health Science and Technology Major Projects (Grant No. 24ZXGZSY00100). D.W.G. acknowledges support from the Ole and Marty Jensen Endowed Chair (University of Utah, Salt Lake City, USA).
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.